- E-mail:BD@ebraincase.com
- Tel:+8618971215294
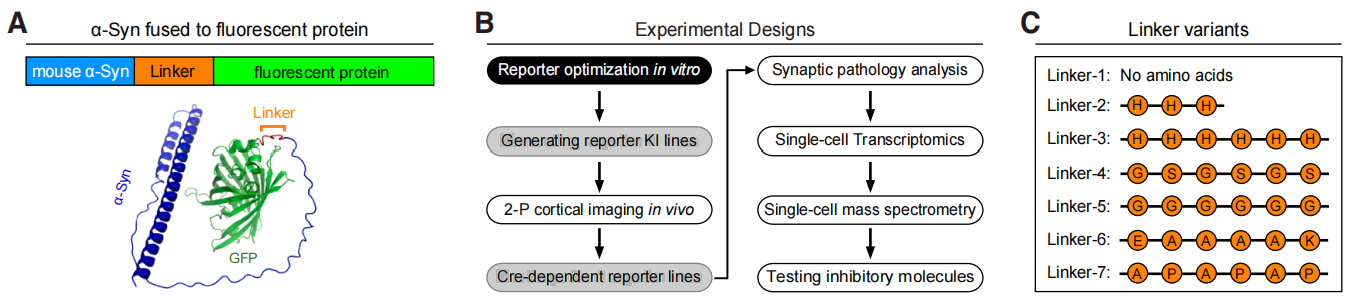
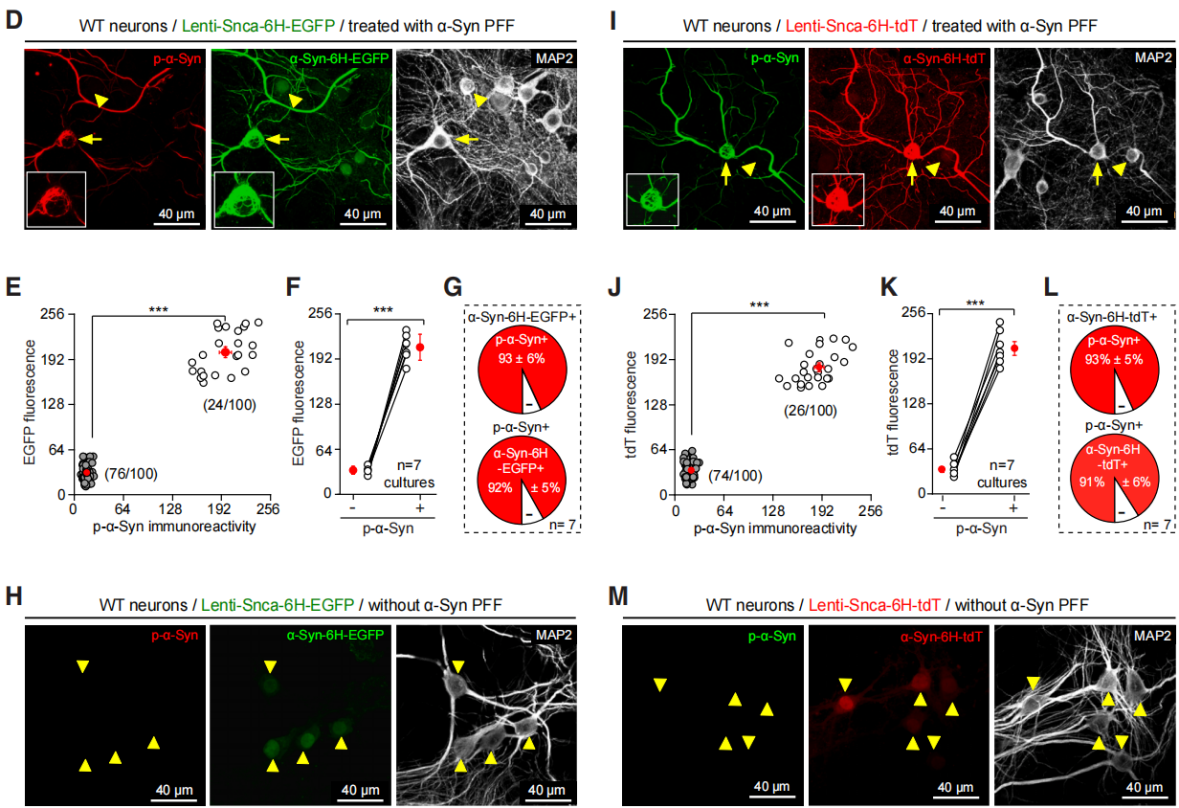
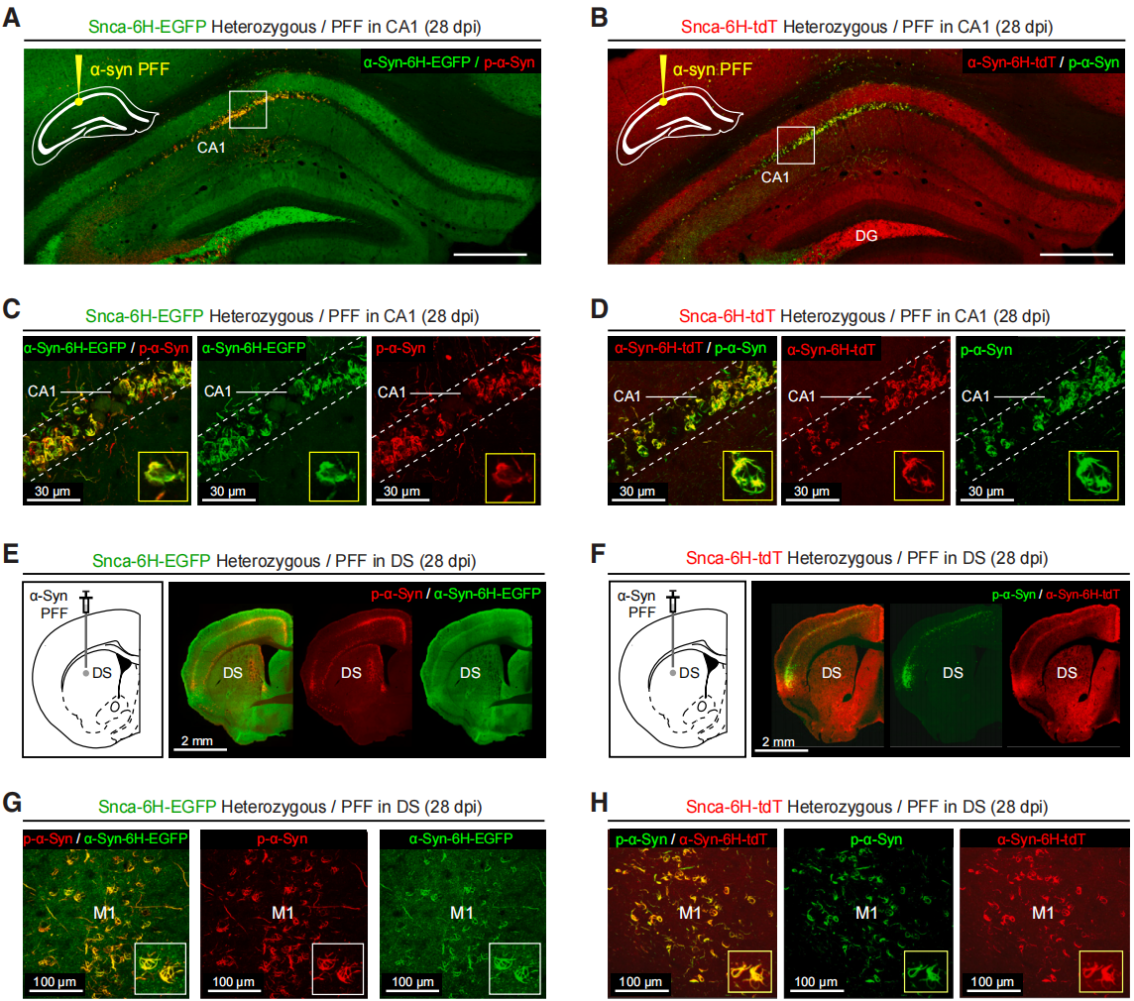
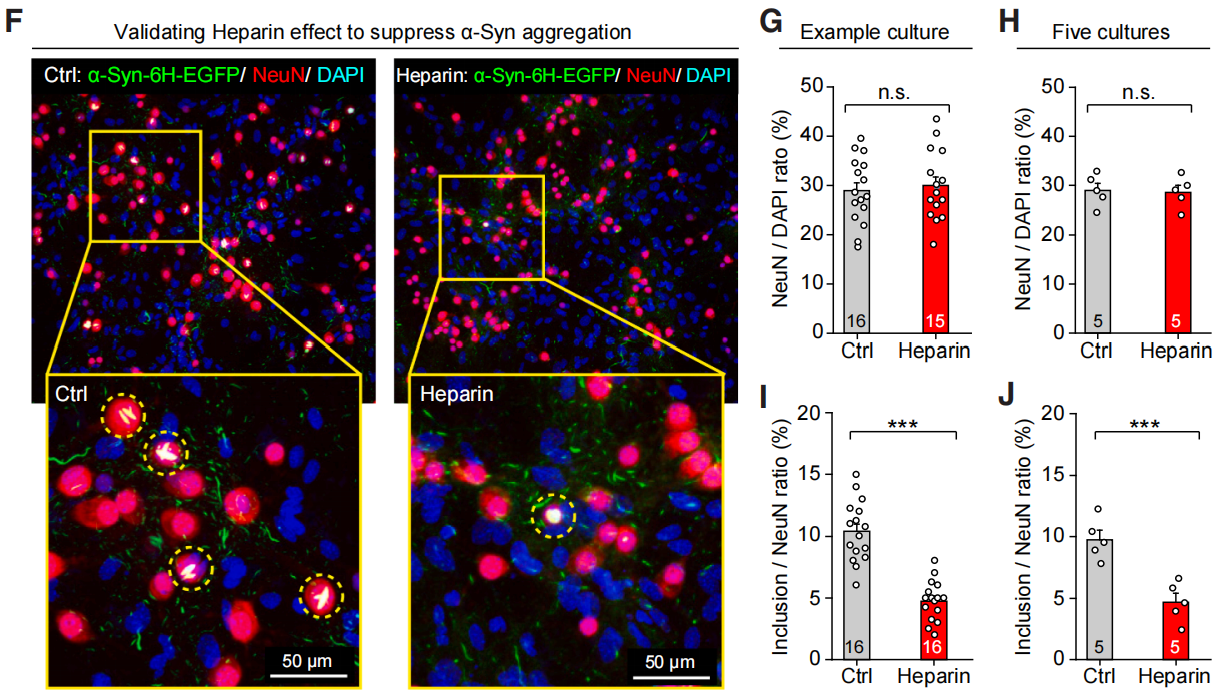
| Virus Type | Product Number | Product Name |
|---|---|---|
| Lentivirus | BM-0827 | rLV-hSyn-mSnca-6H-EGFP-WPRE |
| BM-0828 | rLV-hSyn-mSnca-6H-tdTomato-WPRE | |
| BM-0829 | rLV-hSyn-hSNCA-6H-EGFP-WPRE | |
| BM-0830 | rLV-hSyn-hSNCA-6H-tdTomato-WPRE | |
| Adeno-Associated Virus | BC-6275/9 | rAAV-hSyn-mSnca-6H-EGFP |
| BC-6276/9 | rAAV-hSyn-mSnca-6H-tdTomato | |
| BC-6277/9 | rAAV-hSyn-hSNCA-6H-EGFP | |
| BC-6278/9 | rAAV-hSyn-hSNCA-6H-tdTomato |