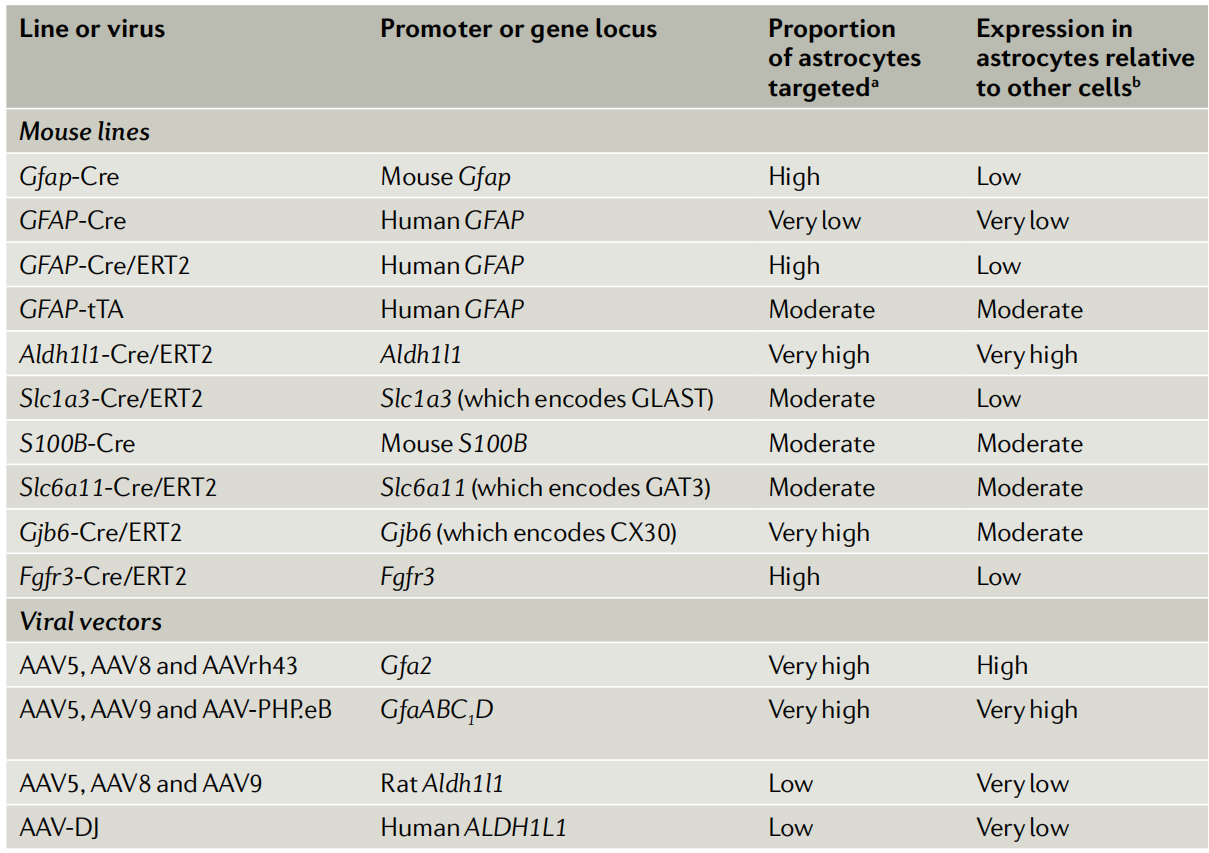
Increasing Astrocyte Ca²⁺ with Channelrhodopsin 2 (ChR2)
ChR2 is a light-controlled non-selective cation channel derived from algae that mediates the influx of protons, sodium, and other cations when stimulated by blue light (~470 nm). When expressed in neurons, it can induce membrane depolarization and action potentials, greatly advancing systems neuroscience. Its use in stimulating astrocyte excitation has been employed to study astrocyte roles in regulating neural circuits and behaviors, including respiration, selective visual cortical responses, blood flow regulation, sleep, and cerebellar motor functions. Research shows that ChR2 stimulation in astrocytes transiently elevates extracellular K⁺ levels, affecting neuronal firing, and this change also impacts astrocytes, microglia, and vasculature. Furthermore, the opsin Arch, which has inhibitory effects in neurons, can enhance Ca²⁺ signaling in cortical astrocytes, suggesting that some tools may have different effects on neurons and astrocytes.
Increasing Astrocyte Ca²⁺ with Melanopsin
Melanopsin is a light-activated GPCR naturally found in retinal ganglion cells and can be activated by blue light (~470–480 nm) and deactivated by yellow light. It almost exclusively induces Gq signaling in neurons. Recently, through AAV expression, melanopsin has been used to selectively stimulate astrocyte Ca²⁺ signaling. Continuous light stimulation of hippocampal astrocytes for 1–20 seconds increases Ca²⁺ signal frequency in their fine processes, with cell body signals triggered only after stimulation longer than 10 seconds. This signal is dependent on IP₃ (disappearing in IP₃R2 knockout mice). Short pulse light stimulation enhances astrocyte Ca²⁺ signaling and simulates short-term potentiation, while low-frequency light stimulation triggers long-term potentiation and enhances contextual memory (consistent with DREADD stimulation results). These findings suggest that melanopsin is a time-controllable light stimulation tool, potentially simulating endogenous oscillatory patterns in GPCR pathways.
Increasing Astrocyte Ca²⁺ with Opto-XRs (Light-Sensitive GPCRs)
Light-controlled GPCR pathways (like opto-XRs) offer faster time control than traditional pharmacological methods, aiding in the assessment of time-locked changes in synaptic function and behavior. Opto-XRs, by replacing the internal loop of vertebrate rhodopsins, can recruit pathways mediated by Gq, Gs, and others, with some having been validated in neurons. In astrocytes, stimulation of opto-α1-ARs replicated the effect of Gq-DREADD stimulation of hippocampal astrocytes in enhancing fear memory. Other opto-XRs have yet to be tested. Limitations of opto-XRs include a limited light propagation range, potential light-induced heating that could affect neurons, and fewer cells being stimulated compared to chemogenetics. However, chemical genetics and opto-α1-AR Gq pathway activation in hippocampal astrocytes have similar effects on fear memory.
Lowering Astrocyte Ca²⁺ via Targeting IP3
Astrocyte Ca²⁺ signaling is complex, with the main pathway being the activation of GPCRs, which leads to IP3 production and subsequent stimulation of IP3 receptors (mainly IP3R2) on the endoplasmic reticulum to release Ca²⁺. In IP3R2 knockout mice, spontaneous and Gq-GPCR-mediated Ca²⁺ signals in astrocytes are significantly weakened, and related studies have been widely reviewed. An IP3 sponge (based on the IP3R1 ligand-binding site) expressed in astrocytes using the GLT1 promoter system reduces hippocampal astrocyte mGluR-mediated Ca²⁺ responses and synaptic coverage, which is associated with hippocampal-dependent behavioral deficits. However, there is a leakage of expression to neurons. The p130PH (IP3 buffer) expressed via AAV with the GfaABC1D promoter inhibits ATP-induced Ca²⁺ elevation in cortical astrocytes. Hypothalamic expression in mice reduces feeding (opposite to Gq-DREADD activation which increases feeding). However, p130PH also binds to other phospholipids, which could affect the interpretation of the results.
Lowering Astrocyte Ca²⁺ via Calcium Exchangers
Astrocyte Ca²⁺ signaling exhibits diversity, and researchers have developed genetic methods independent of Ca²⁺ sources. By overexpressing the efficient calcium pump PMCA2w/b, which is not endogenously expressed in astrocytes, on the surface of striatal astrocytes controlled by the GfaABC1D promoter through AAV, they were able to reduce the amplitude and duration of spontaneous and GPCR-mediated Ca²⁺ signals by about 80%, with no significant change in frequency. In adult mice, reducing Ca²⁺ signals in dorsal-lateral striatal astrocytes leads to repetitive behavior (similar to human obsessive-compulsive disorder symptoms). The mechanism is through the upregulation of the GABA transporter GAT3 in astrocytes, reducing tonic GABA inhibition on striatal neurons. RNA-seq showed that reduced Ca²⁺ levels led to decreased expression of related genes, enhancing GAT3 function. Notably, PMCA2w/b did not significantly change extracellular Ca²⁺ concentrations and did not affect neurotransmitter release probability.
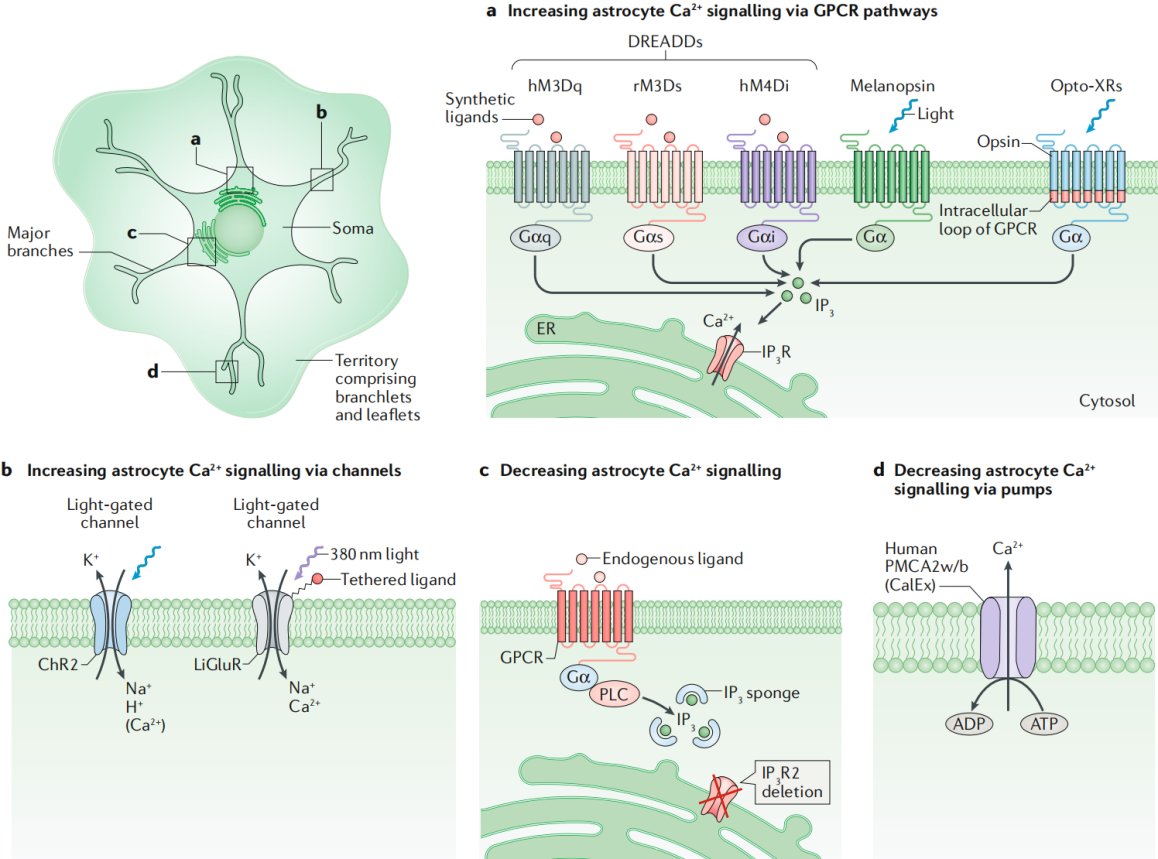 Figure 4: Schematic of Tools for Manipulating Astrocyte Intracellular Calcium Signaling
Figure 4: Schematic of Tools for Manipulating Astrocyte Intracellular Calcium Signaling
Conclusion
Using currently available methods and technologies, experiments can be designed to explore astrocyte biology from molecular to system levels. However, more methods are needed to explore how various cells in the nervous system— including astrocytes, neurons, and microglia—interact in health, neuroimmunity, and brain-periphery interactions. Full control over models, methods, and technologies is critical for the advancement of neuroglial biology and may lead to significant breakthroughs in cell biology and medicine.
BrainCase Biotech offers a variety of custom and viral packaging services.
For more information and service details,
please contact bd@ebraincase.com