- E-mail:BD@ebraincase.com
- Tel:+8618971215294
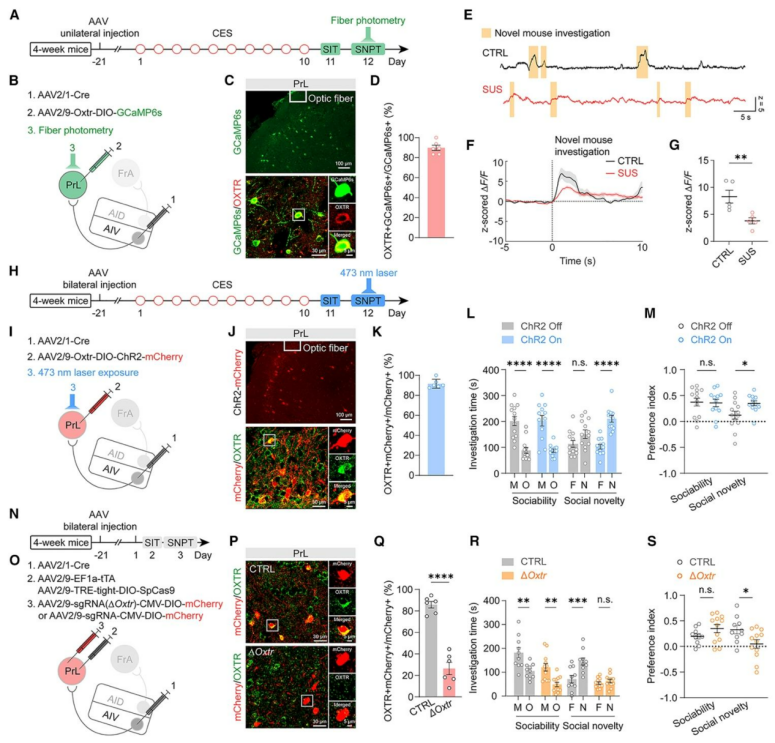
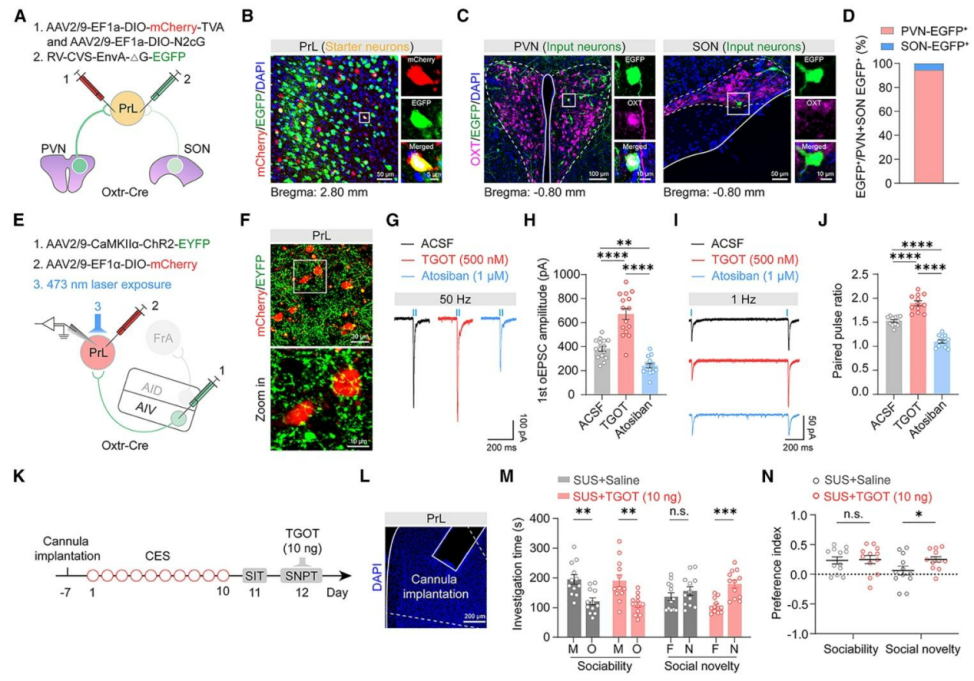
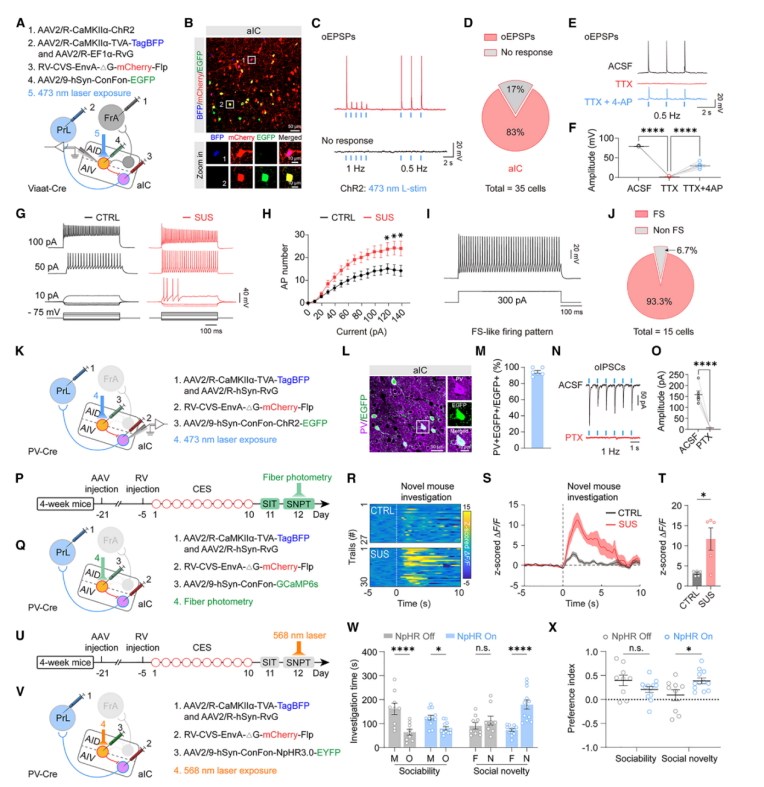
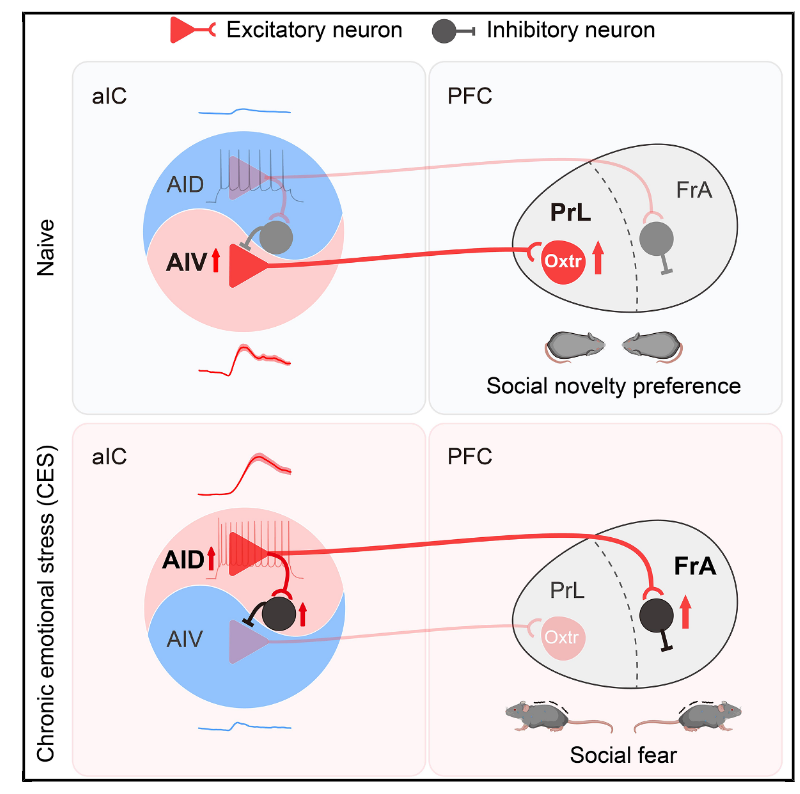
| Product Category | Product Number | Product Name |
|---|---|---|
| Fluorescent Proteins | BC-0016 | rAAV-EF1α-DIO-mCherry |
| BC-0023 | rAAV-hSyn-mCherry | |
| BC-0020 | rAAV-hSyn-EGFP | |
| BC-0028 | rAAV-CaMKIIα-mCherry | |
| BC-0027 | rAAV-CaMKIIα-EGFP | |
| BC-0193 | rAAV-EF1α-fDIO-mCherry | |
| BC-0015 | rAAV-EF1α-DIO-EGFP | |
| BC-0029 | rAAV-CaMKIIα-EYFP | |
| BC-3848 | rAAV-hSyn-Con Fon-EGFP | |
| Recombinases | BC-0243/1 | rAAV-CMV-Cre |
| BC-0171 | rAAV-hSyn-Flp | |
| BC-0165 | rAAV-CaMKIIα-EGFP-P2A-SV40 NLS-Cre | |
| Chemogenetics | BC-0155 | rAAV-EF1α-DIO-hM4D(Gi)-mCherry |
| BC-0146 | rAAV-EF1α-DIO-hM3D(Gq)-mCherry | |
| Optogenetics | BC-0356 | rAAV-hSyn-ConFon-hChR2(H134R)-P2A-EYFP |
| BC-0120 | rAAV-CaMKIIα-eNpHR3.0-EYFP | |
| BC-0100 | rAAV-CaMKIIα-hChR2(H134R)-EYFP | |
| BC-1134 | rAAV-Oxtr-DIO-ChR2-mCherry | |
| Calcium Imaging | BC-0081 | rAAV-CaMKIIα-GCaMP6s |
| BC-2016 | rAAV-Ef1α-ConFon-GCaMP6s | |
| RV-helper | BC-0061 | rAAV-EF1α-DIO-mCherry-T2A-TVA |
| BC-0442 | rAAV-EF1α-DIO-N2cG | |
| RV | BC-RV-EnvA862 | RV-EnvA-ΔG-EGFP |
| BC-RV-CVS EnvA471 | CVS-EnvA-ΔG-mCherry-P2A-FlpO | |
| Tetracycline-Inducible Gene Regulation | BC-0055 | rAAV-EF1α-tTA |
| BC-2256 | rAAV-TRE-tight-DIO-SpCas9-3×FLAG | |
| BC-2255 | AAV-U6-sgRNA(ΔOxtr)-CMV-DIO-mCherry | |
| BC-1629 | rAAV-U6-sgRNA-CMV-DIO-mCherry |