- E-mail:BD@ebraincase.com
- Tel:+8618971215294
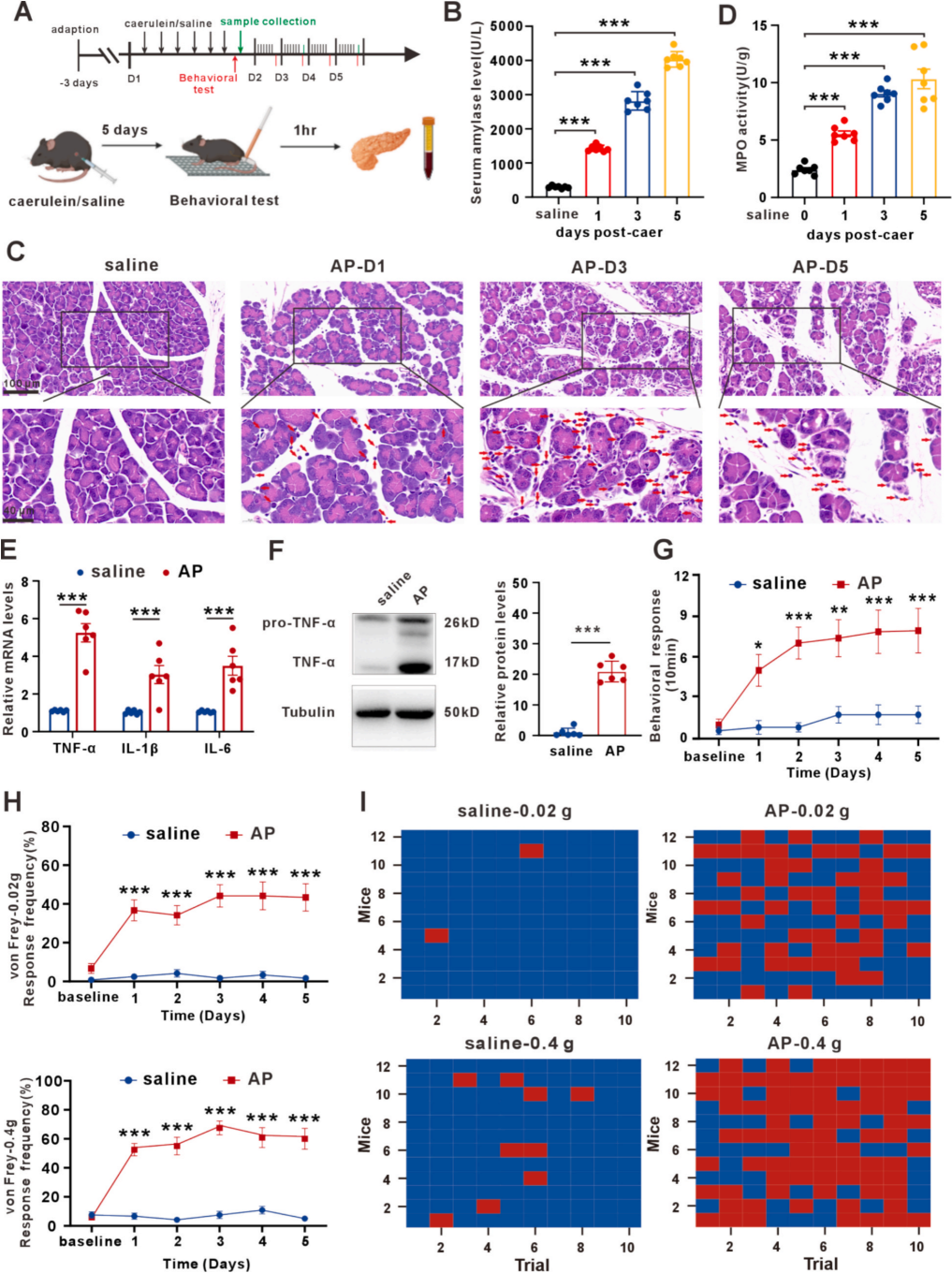
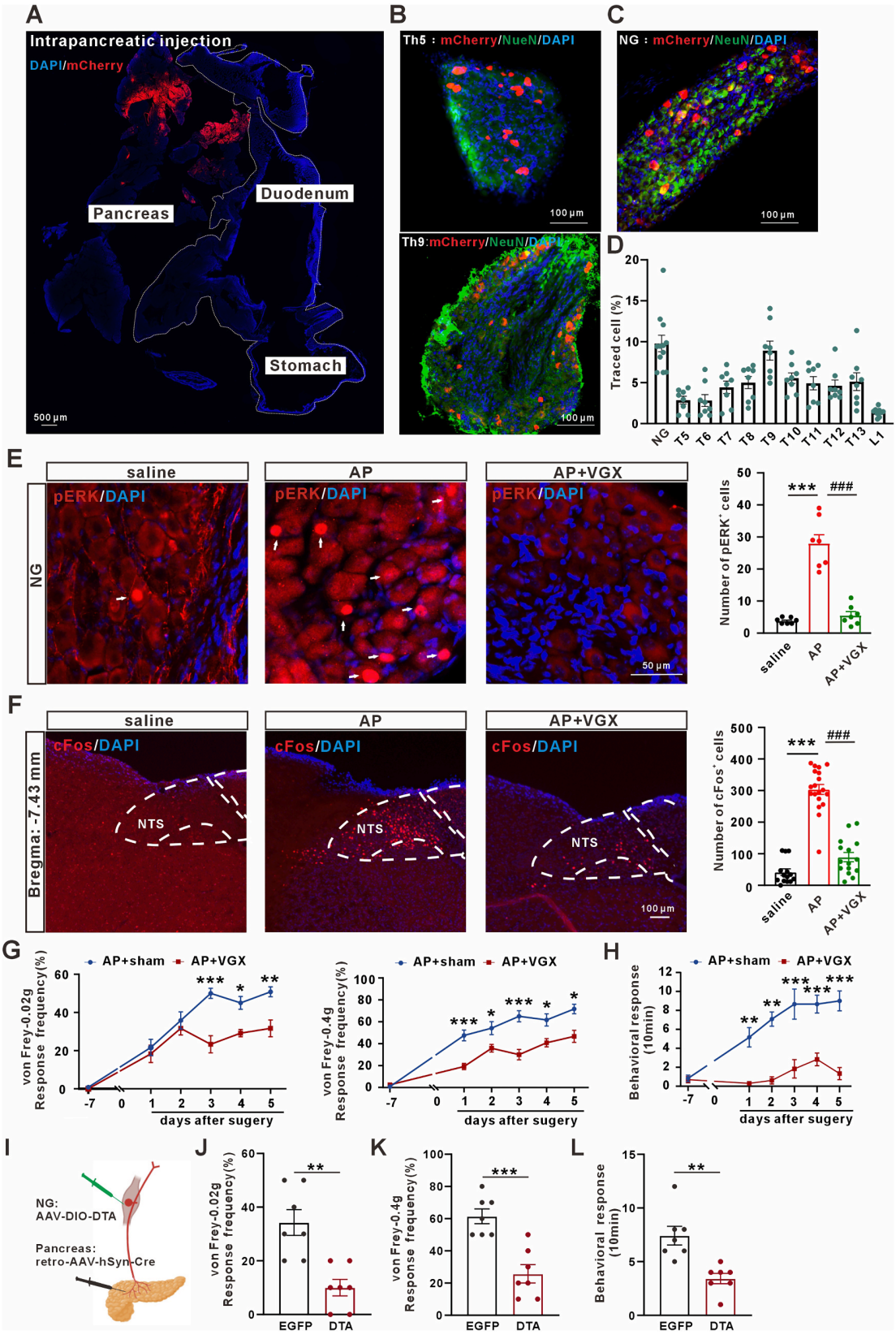
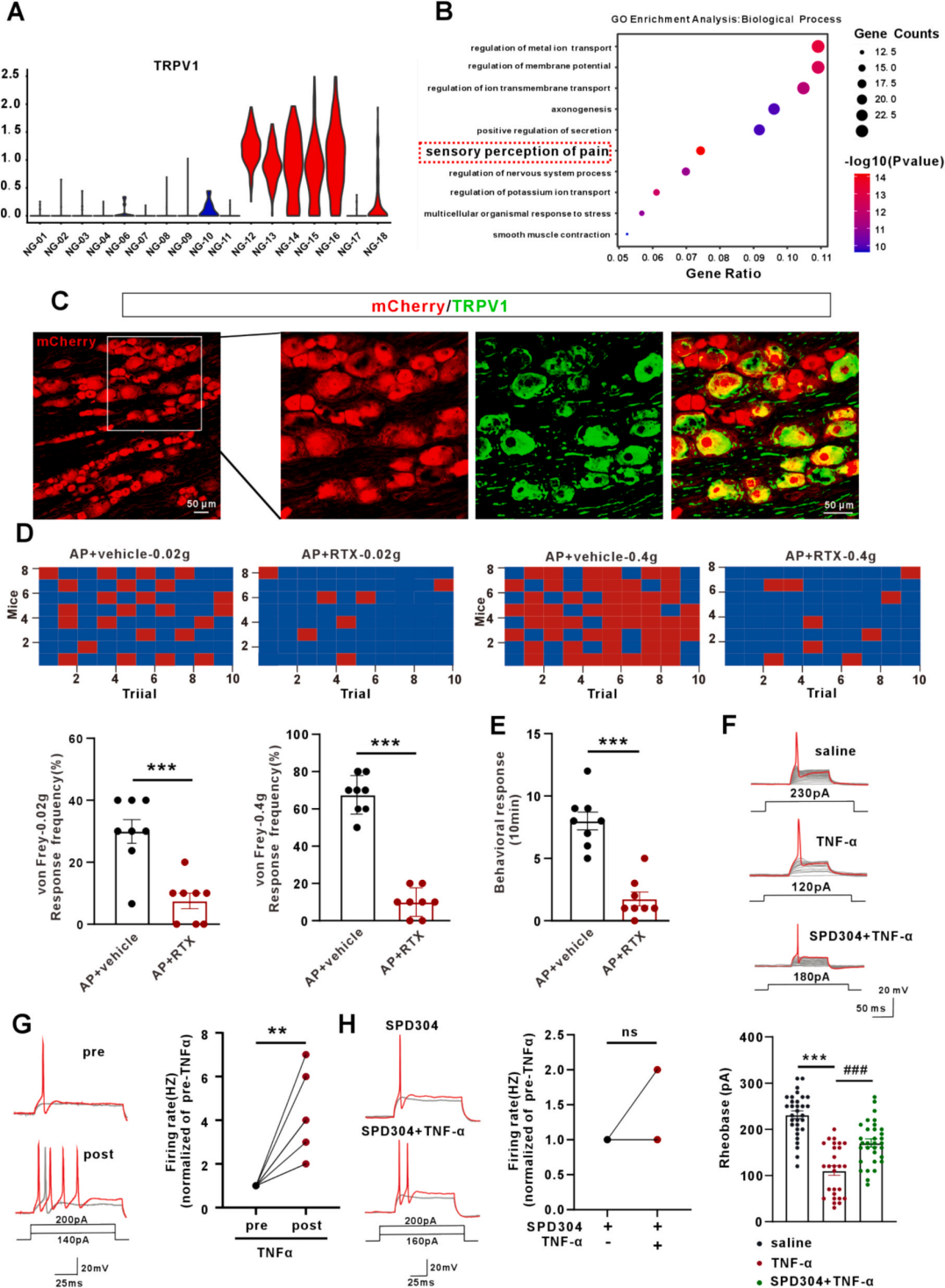
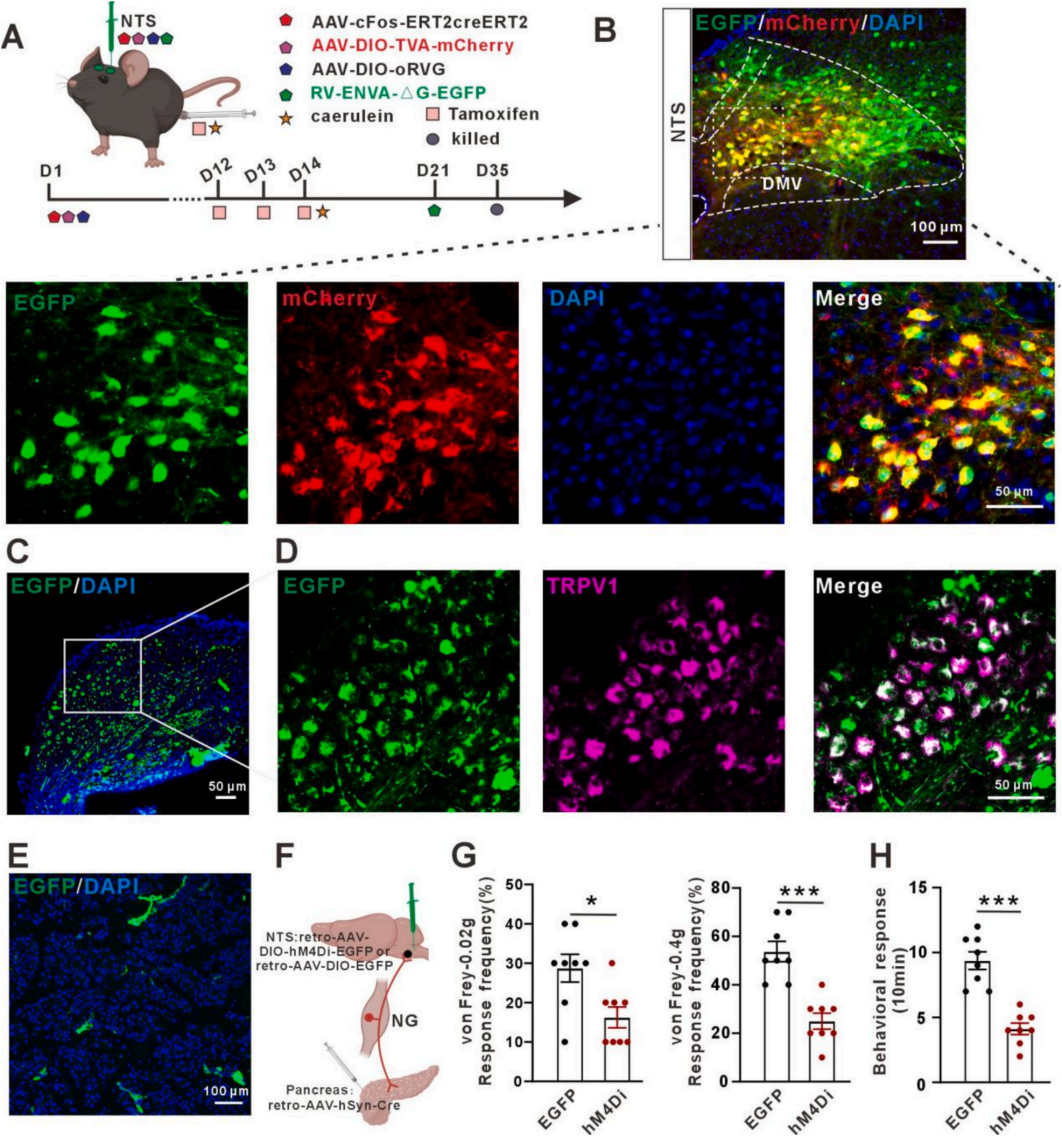
| Product Category | Product Number | Product Name |
|---|---|---|
| Recombinase | BC-0164 | rAAV-CaMKIIα-Cre |
| Chemogenetics | BC-0146 | rAAV-EF1α-DIO-hM3D(Gq)-mCherry |
| BC-0154 | rAAV-EF1α-DIO-hM4D(Gi)-EYFP | |
| Optogenetics | BC-0126 | rAAV-EF1α-DIO-eNpHR3.0-mCherry |
| Calcium Imaging | BC-0086 | rAAV-EF1α-DIO-GCaMP6s |
| BC-0081 | rAAV-CaMKIIα-GCaMP6s | |
| Fluorescent Proteins | BC-0023 | rAAV-hSyn-mCherry |
| BC-0027 | rAAV-CaMKIIα-EGFP | |
| BC-0244 | rAAV-hSyn-DIO-EGFP | |
| BC-1168 | rAAV-cFos-Cre-ERT2-P2A-EGFP | |
| Apoptosis | BC-0132 | rAAV-EF1α-DIO-DTA |
| RV Retroviral Transfection System | BC-0442 | rAAV-EF1α-DIO-N2cG |
| BC-0061 | rAAV-EF1α-DIO-mCherry-F2A-TVA | |
| BC-RV-CVS EnvA461 | CVS-EnvA-ΔG-EGFP |