- E-mail:BD@ebraincase.com
- Tel:+8618971215294
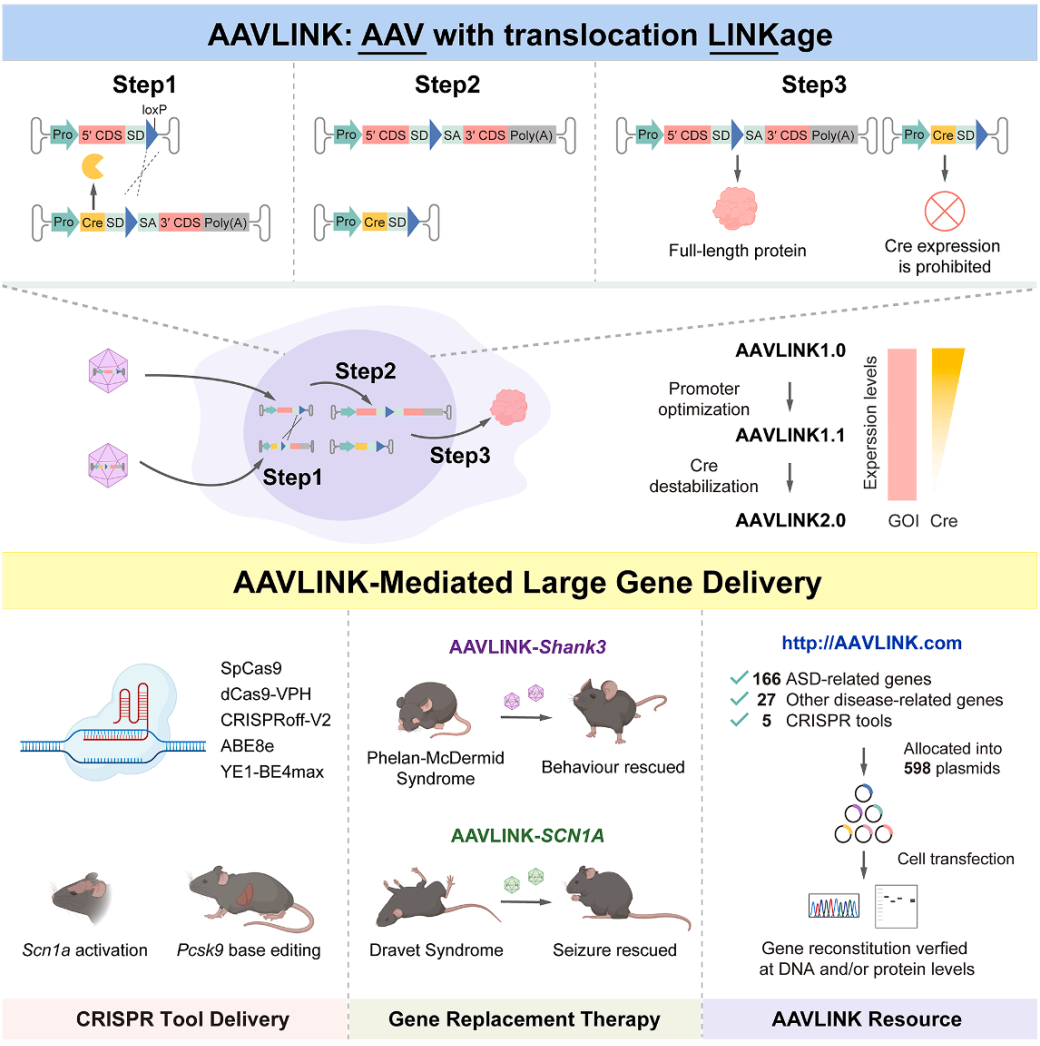
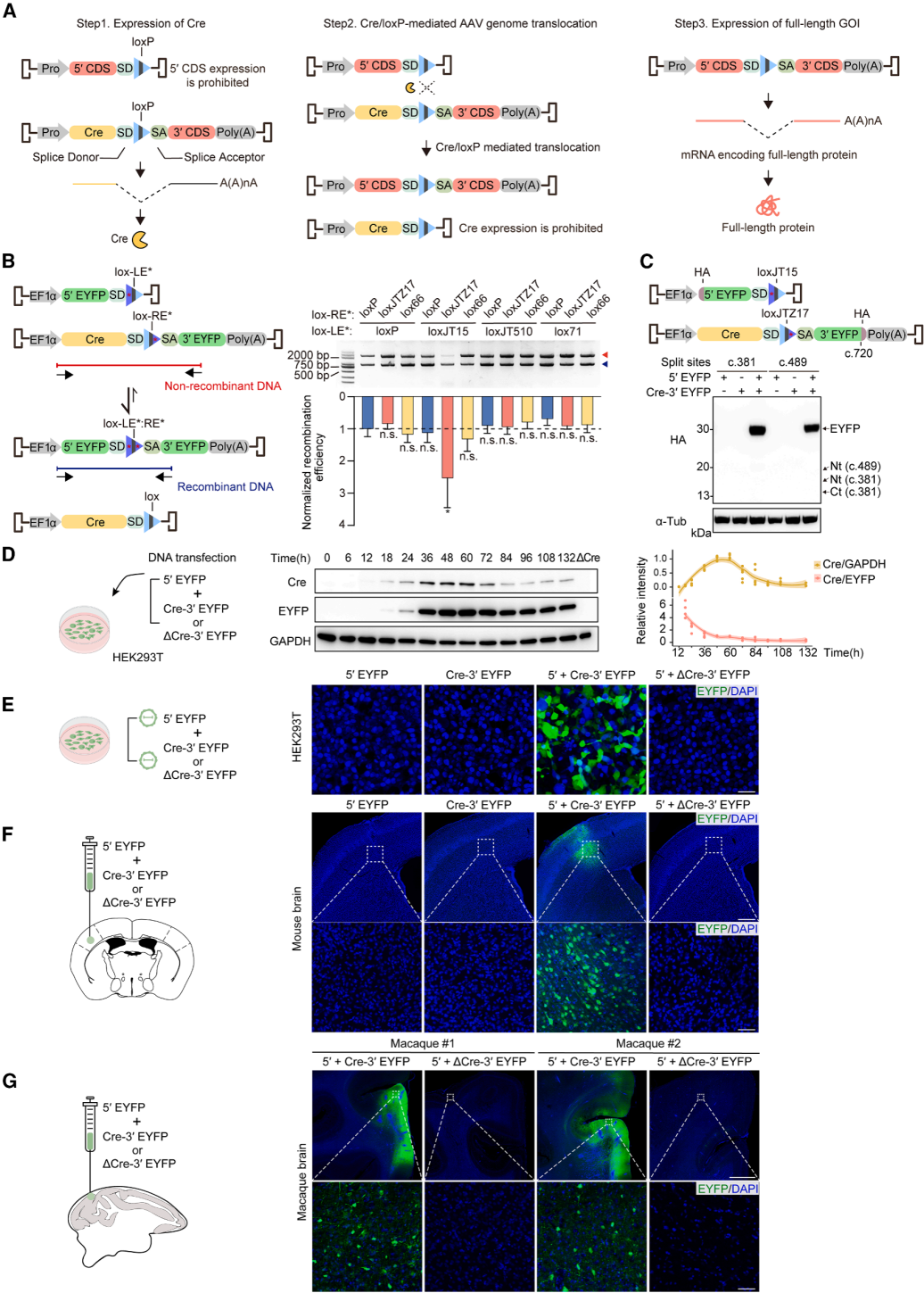
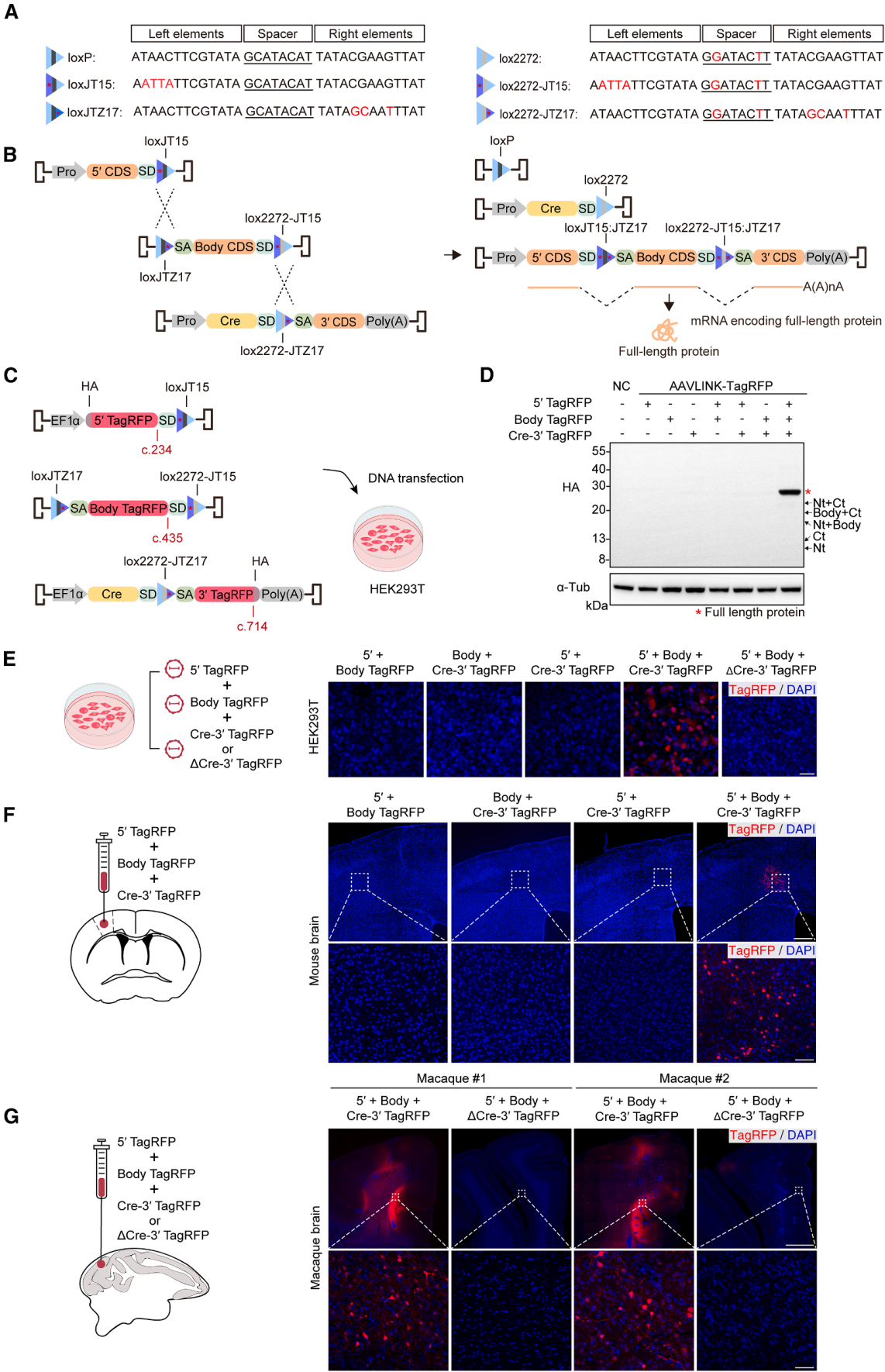
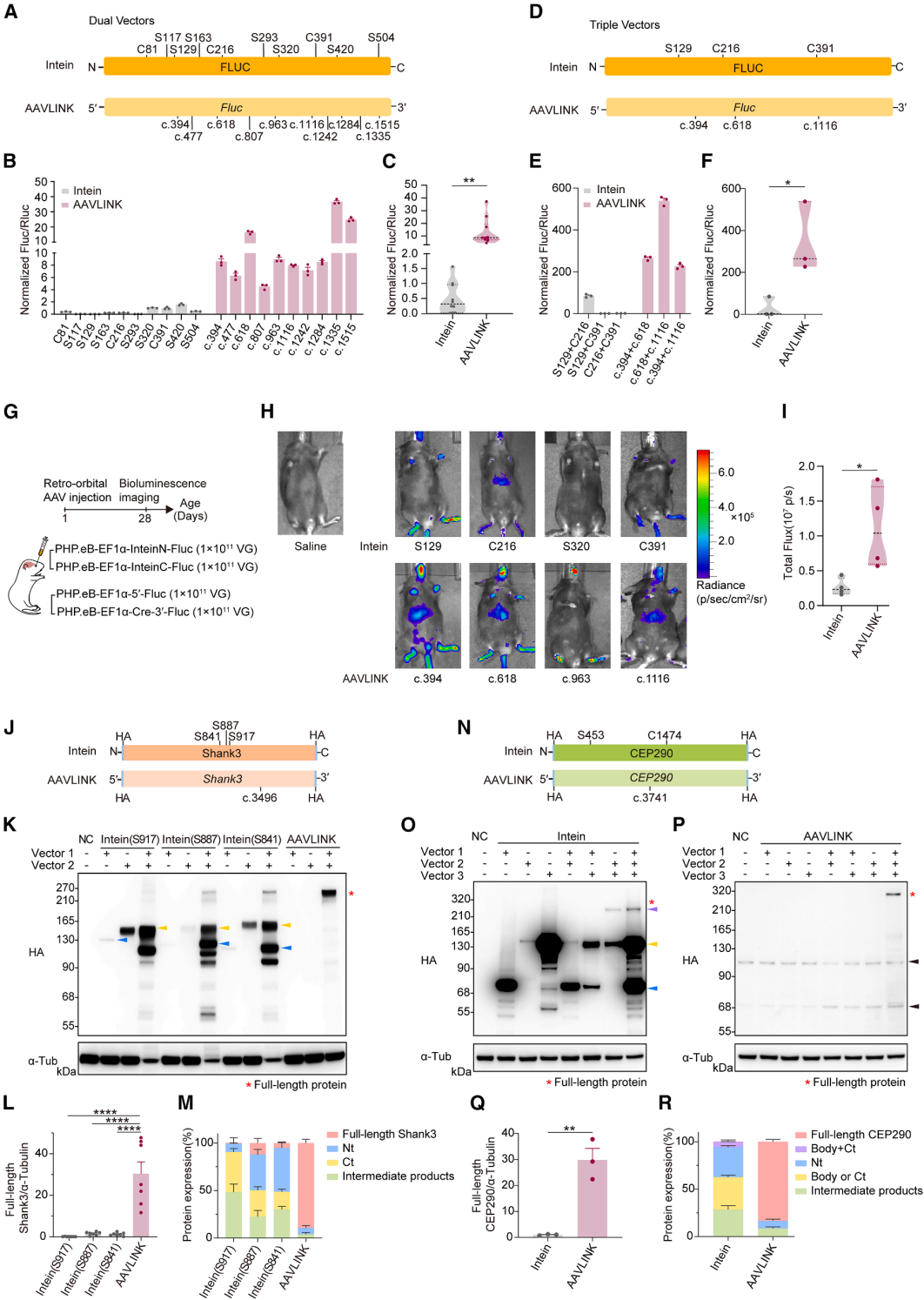
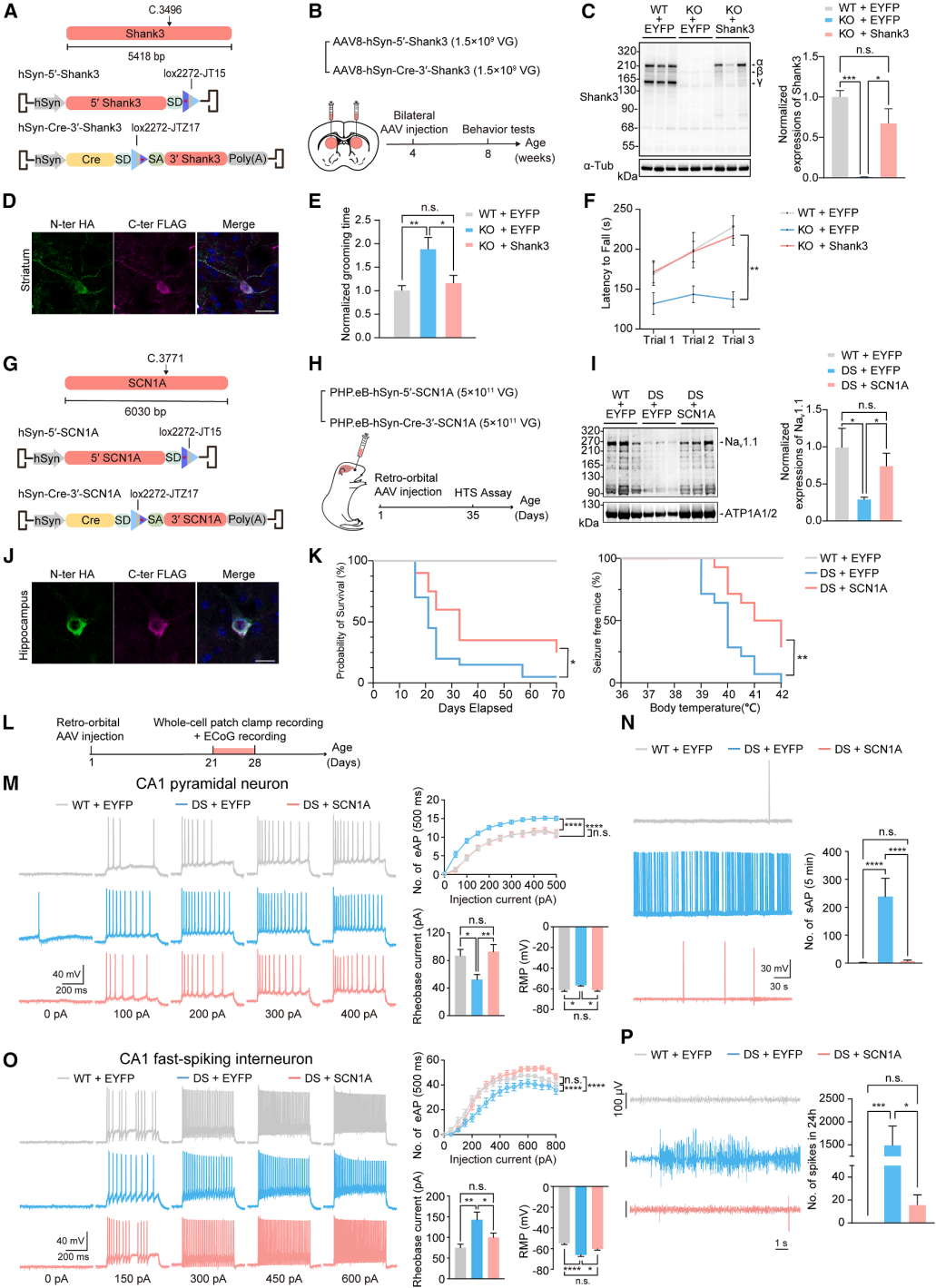
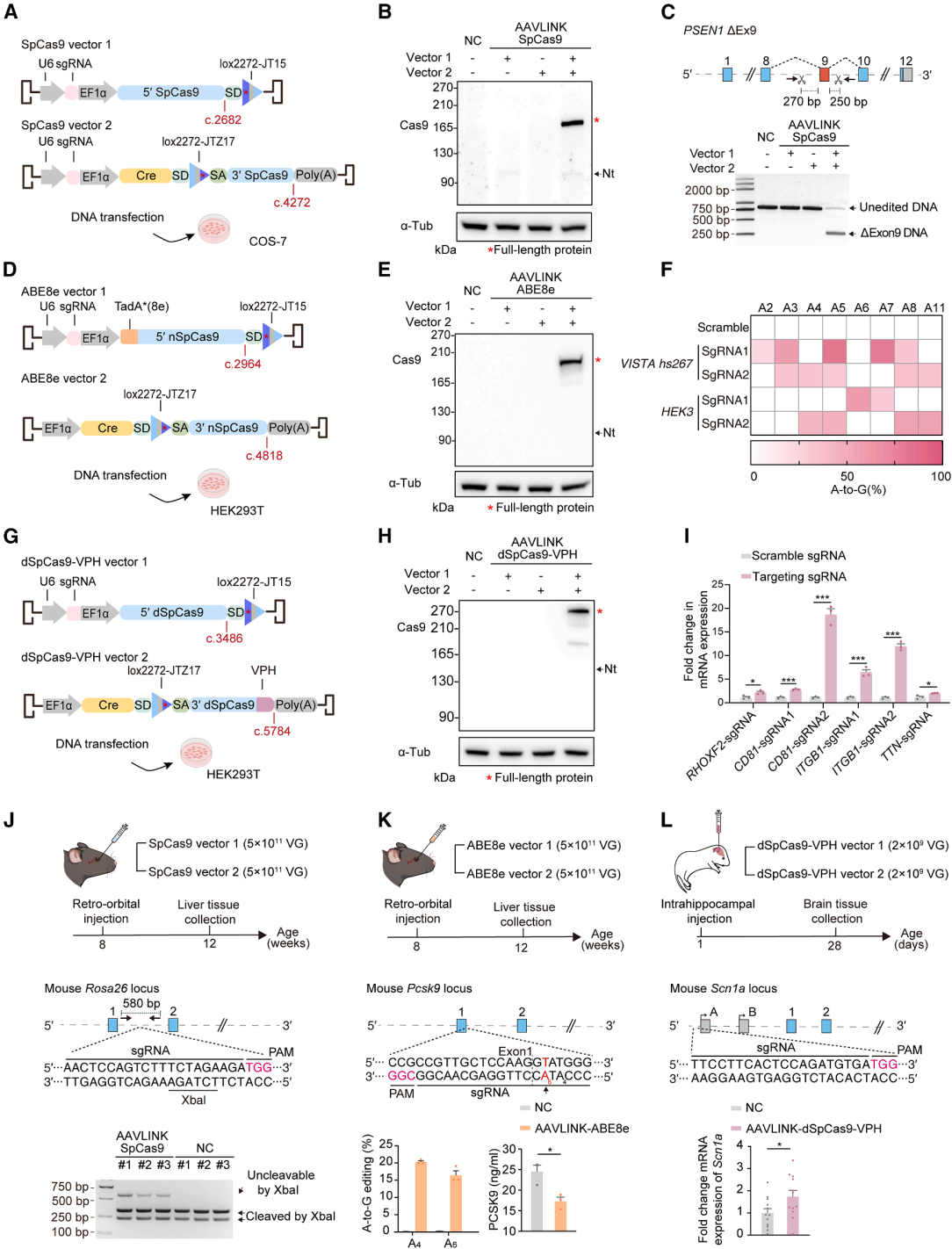
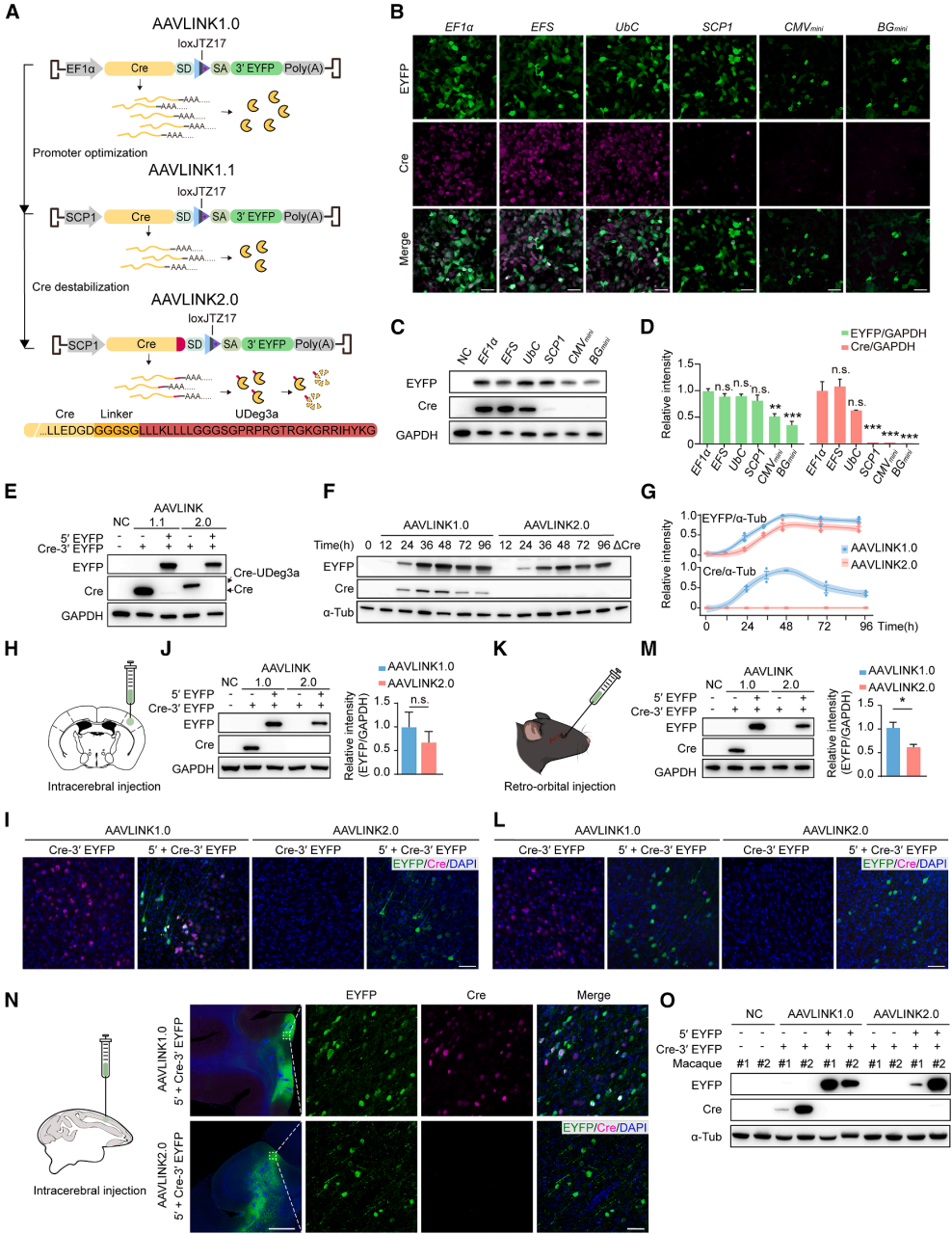
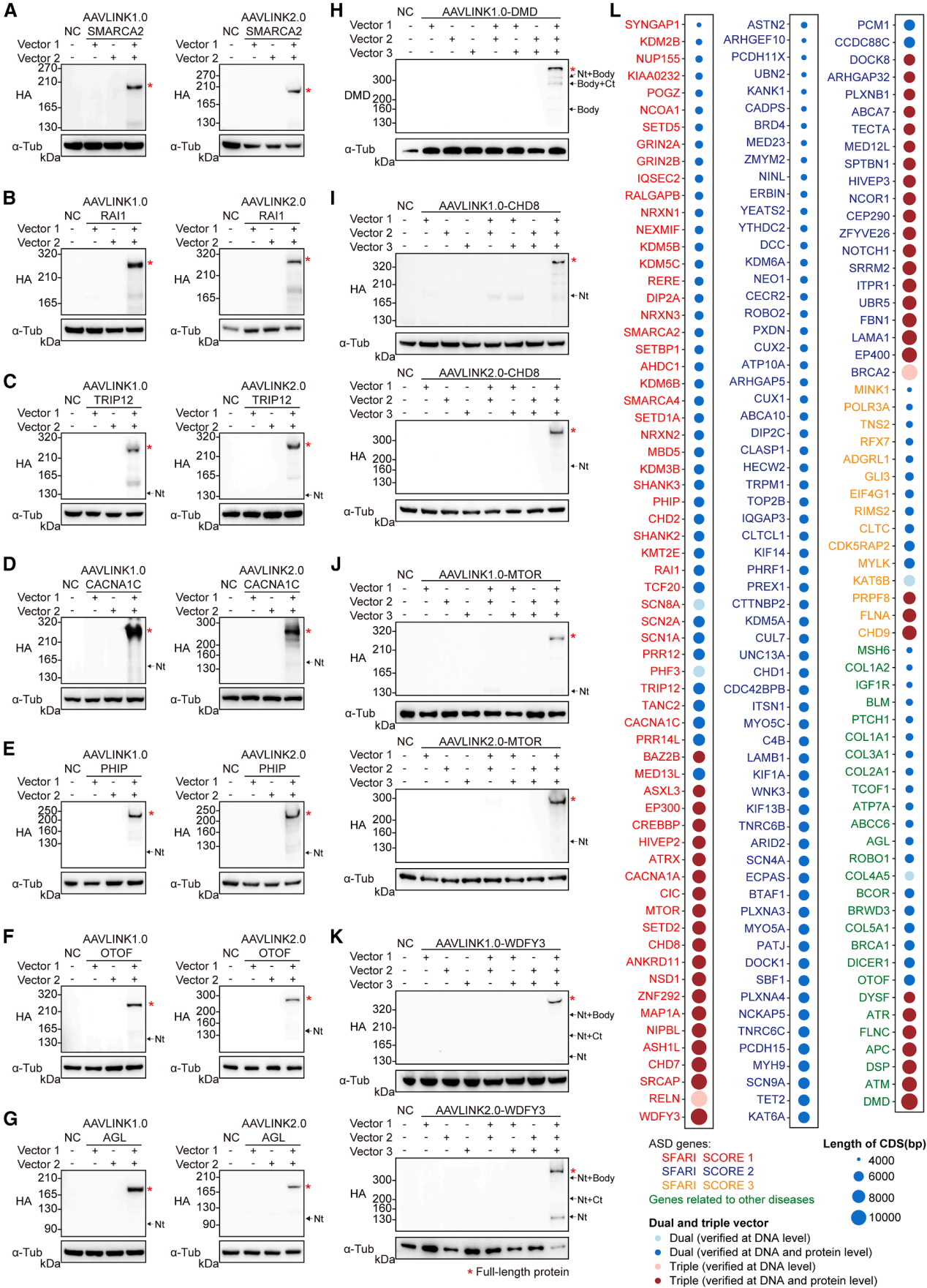
Exogenous Cre recombinase presents a potential biosafety risk. Even though the expression of Cre in AAVLINK is transient (Figure 1D), further optimization was made to reduce its expression (Figure 6A).
Step 1: Screening Weak Promoters (AAVLINK1.1)
After testing six promoters (EF1α, EFS, UbC, SCP1, CMVmini, BGmini), Western Blot (WB) and Immunohistochemistry (IHC) results showed that the SCP1 promoter efficiently maintained high recombination of EYFP while minimizing Cre expression (Figure 6B-6D). Based on this, AAVLINK1.1 was developed (Figure 6A).
Step 2: Adding Unstable Tags (AAVLINK2.0)
The C-terminal of Cre was fused with a potent protein instability tag, UDeg3a, which rendered Cre nearly undetectable without affecting recombination efficiency (Figure 6E-6G). This resulted in the final version, AAVLINK2.0 (Figure 6A).
Comparing Cre expression in vivo through intracranial and intravenous injection, the two versions of AAVLINK were tested:
AAVLINK1.0:
After dual-vector delivery, IHC detected weak Cre expression, and WB showed no Cre detection, confirming that Cre was inactivated (Figure 1A, 6I, 6J, 6L, 6M).
AAVLINK2.0:
Regardless of the delivery method, both IHC and WB failed to detect Cre expression, and the target gene was efficiently expressed (Figure 6I, 6J, 6L, 6M).
Additionally, experiments in macaques verified that AAVLINK2.0 had no Cre expression and efficiently expressed the target gene (Figure 6N, 6O). AAVLINK2.0, optimized with a "weak promoter + protein degradation tag" combination, resolved the biosafety concerns of Cre expression and enhanced its clinical translation potential.