- E-mail:BD@ebraincase.com
- Tel:+8618971215294
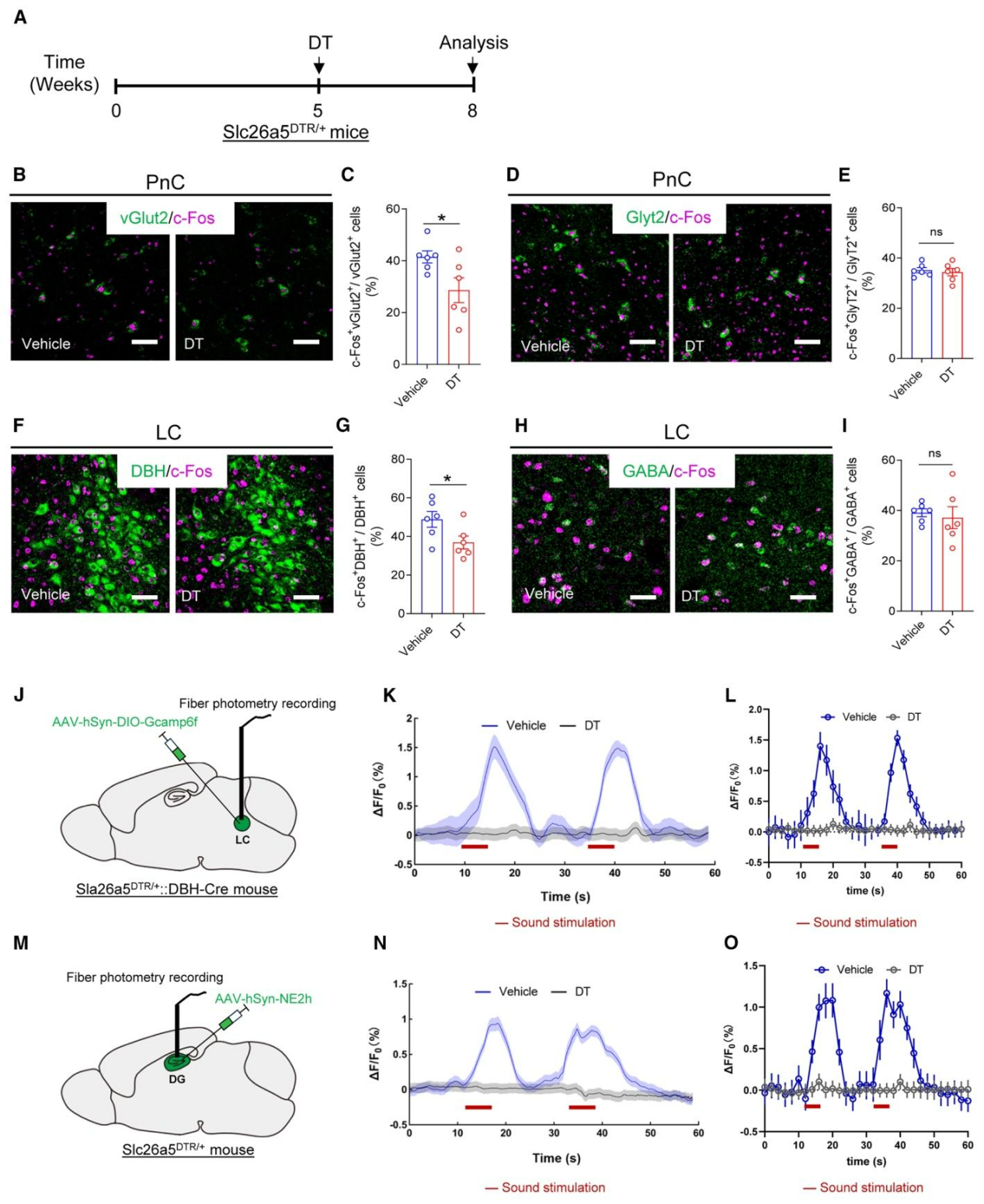
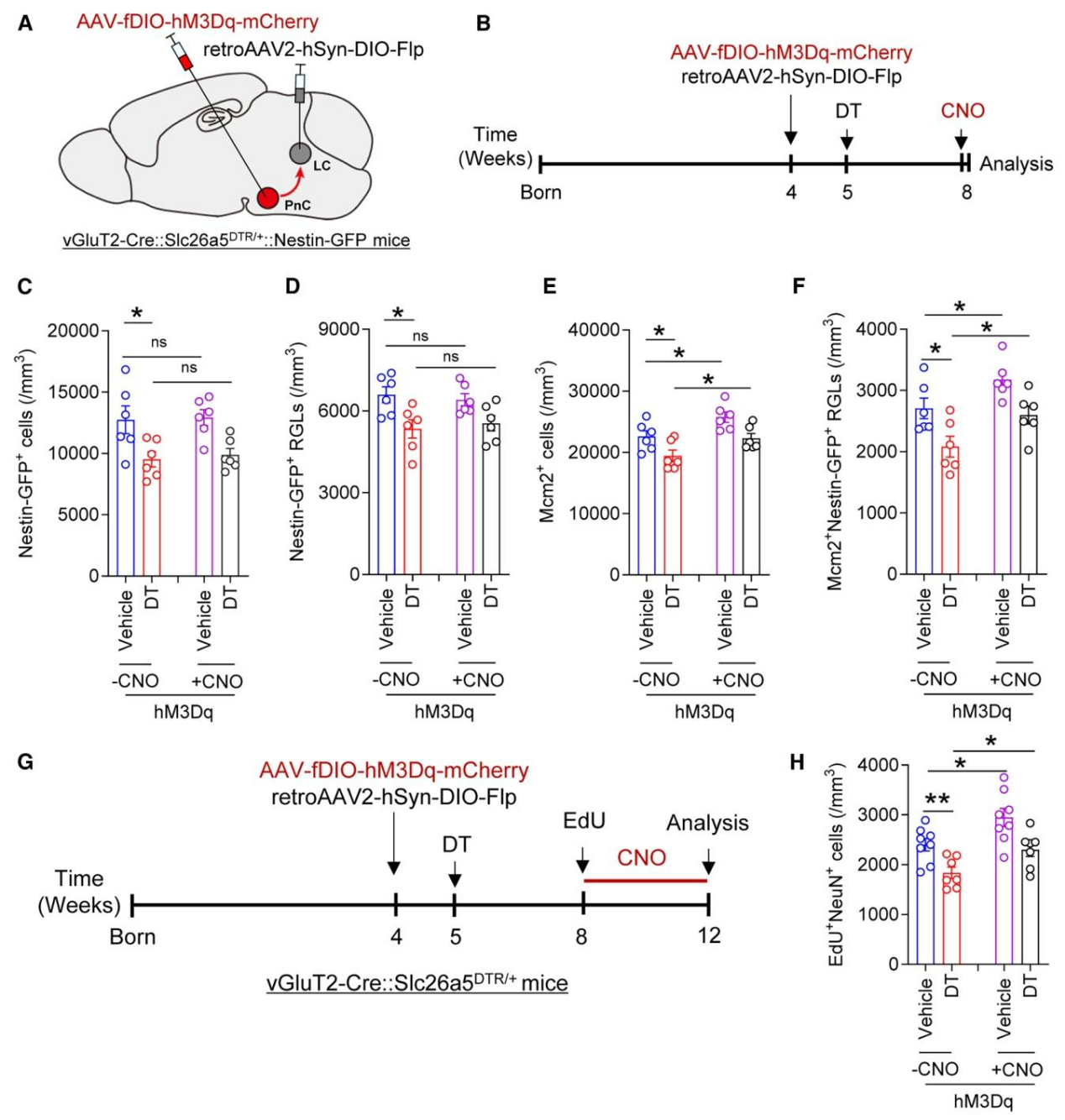
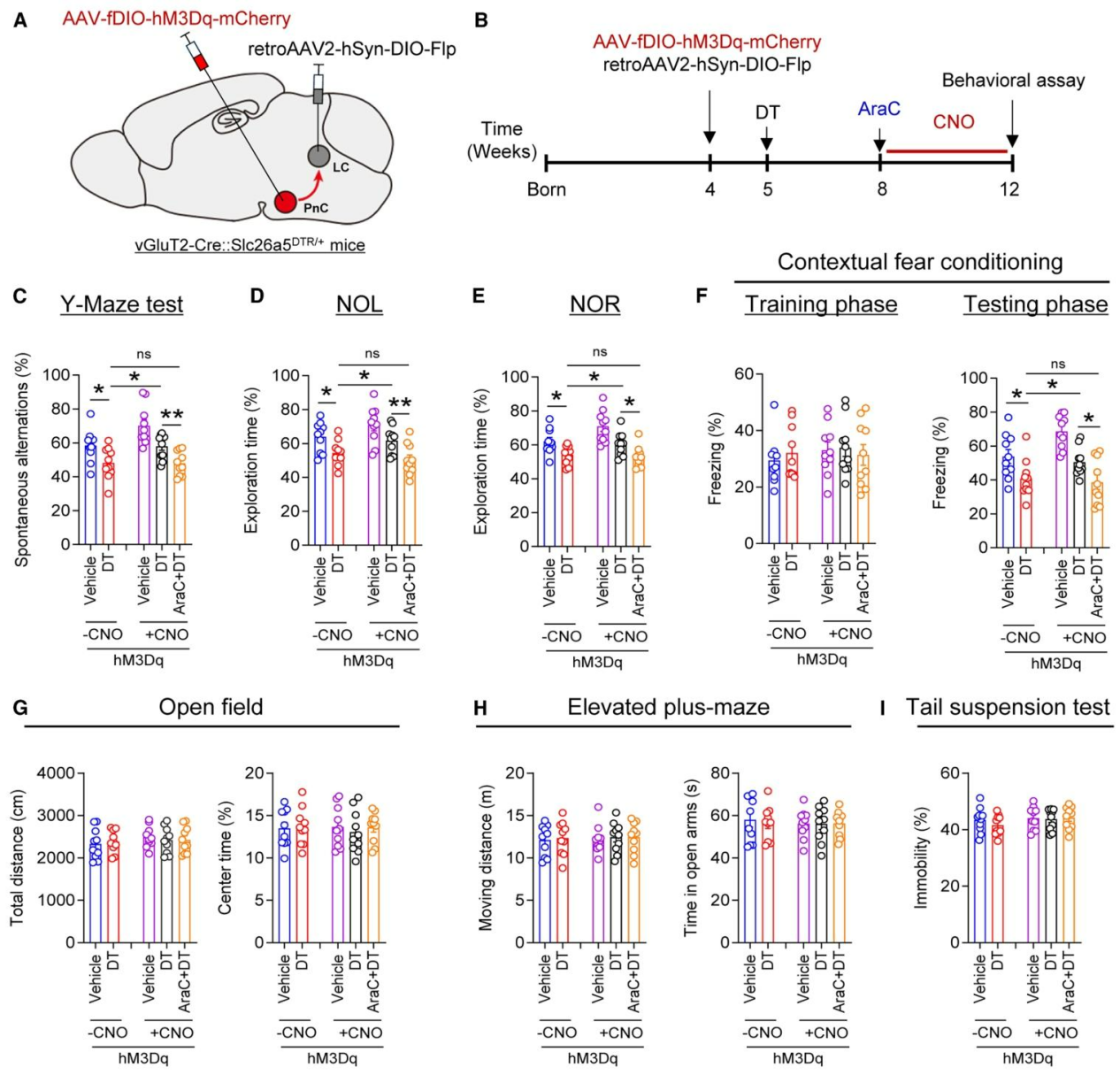
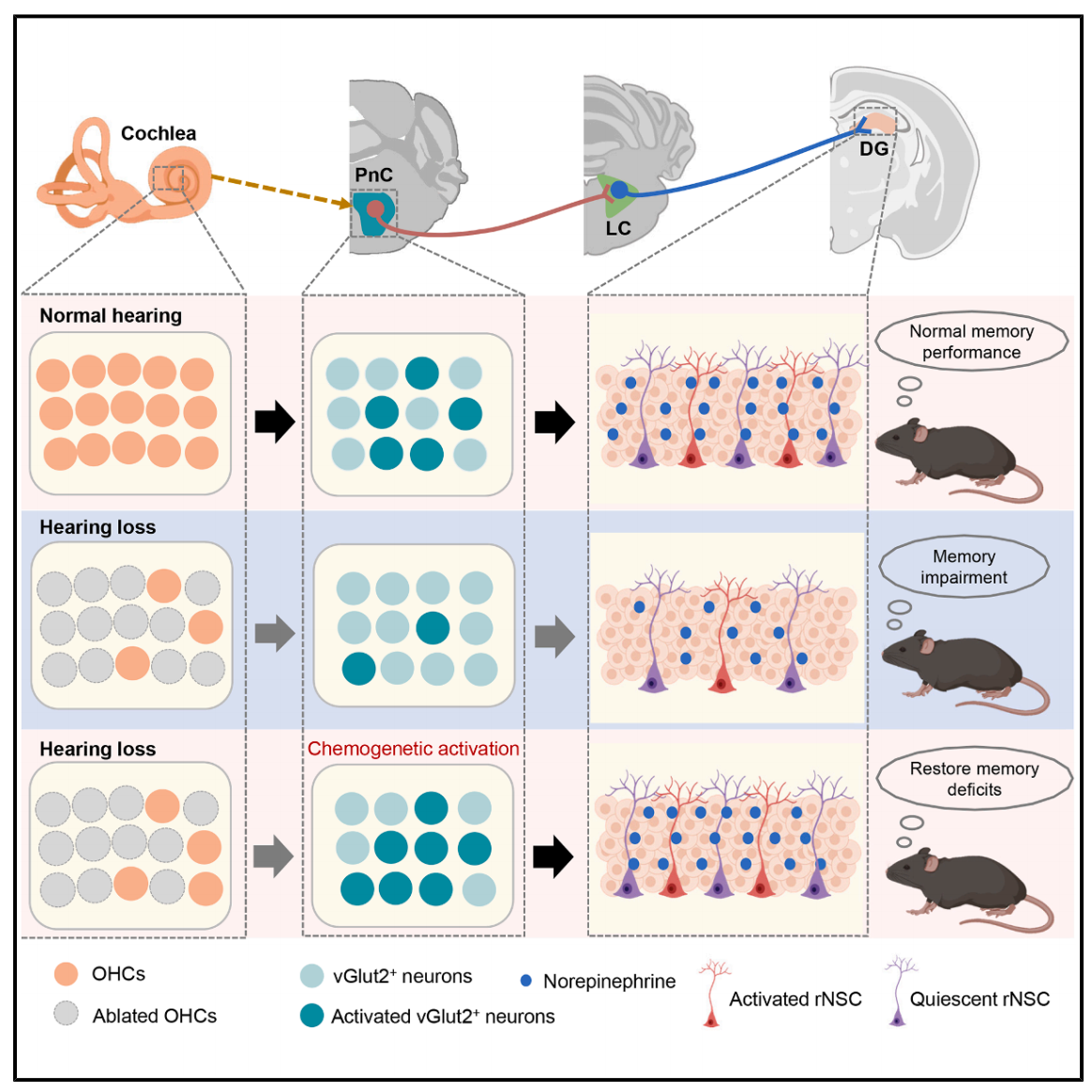
| Product Category | Product Number | Product Name |
|---|---|---|
| Fluorescent Protein | BC-0025 | rAAV-hSyn-DIO-mCherry |
| BC-0023 | rAAV-hSyn-mCherry | |
| BC-0020 | rAAV-hSyn-EGFP | |
| BC-0244 | rAAV-hSyn-DIO-GFP | |
| Recombinase | BC-0172 | rAAV-hSyn-SV40 NLS-Flp |
| BC-0178 | rAAV-EF1α-DIO-Flp | |
| BC-0159 | rAAV-hSyn-SV40 NLS-Cre | |
| Chemogenetics | BC-5388 | rAAV-hSyn-FDIO-hM4D(Gi)-P2A-mCherry |
| BC-4927 | rAAV-hSyn-FDIO-hM3D(Gq)-P2A-mCherry | |
| Optogenetics | BC-0101 | rAAV-CaMKIIα-hChR2(H134R)-mCherry |
| BC-4623 | rAAV-hSyn-DIO-eNpHR3.0-P2A-mCherry | |
| Calcium Imaging | BC-0088 | rAAV-EF1α-DIO-GCaMP6f |
| Fluorescent Probe | BC-0268 | rAAV-hSyn-NE2h |
| Retrograde Monosynaptic | BC-0041 | rAAV-EF1α-DIO-EGFP-T2A-TVA |
| BC-0442 | rAAV-EF1α-DIO-N2cG | |
| BC-RV-EnvA844 | RV-EnvA-ΔG-mCherry | |
| Anterograde Monosynaptic | BC-0045 | rAAV-EF1α-DIO-EGFP-T2A-TK(HSV) |
| BC-HSVΔTK-tdT | H129ΔTK-hUbC-tdTomato |